Chaperonin GroEL
Chaperonin GroEL: a cradle of proteins
【Chaperonin family】
Chaperonin is one of best-studied molecular chaperones. Chaperonin GroEL is an essential chaperone that assists in protein folding in the cell. (Chaperone and chaperonin are often confusing, but chaperone is a general protein family. Conserved chaperone includes Hsp70/DnaK, Hsp90, Hsp104/ClpB, and chaperonin (GroEL/CCT). CCT). Chaperonin is one class of chaperone family.)
【GroEL: Bacterial chaperonin】
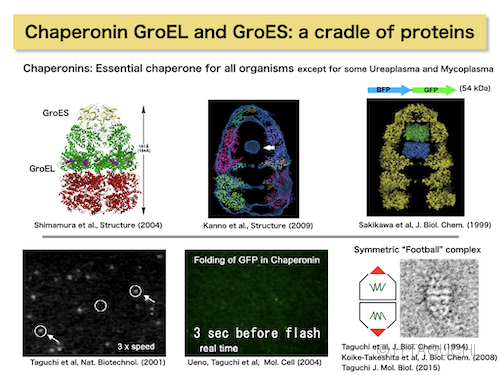
The best-characterized chaperonin is Escherichia coli GroEL and its cochaperonin GroES. The GroEL subunit forms a heptameric ring, which stacks back-to-back to form a double-ring structure, and requires the dome-shaped heptameric GroES. GroEL assists in the folding of a wide variety of substrate proteins in the cell in an ATP-dependent manner, with the aid of GroES.
【Our interests】
-Molecular mechanism of the GroEL-GroES cycling
-Identification of GroEL substrate proteins in the cell
-How GroEL discriminates in vivo substrate proteins
【Selected publications】
-*Taguchi H.,
Reaction cycle of chaperonin GroEL via symmetric “football” intermediate (review).
J Mol Biol 427, 2912-2918 (2015)
-Ishimoto T, Fujiwara K, Niwa T., *Taguchi H.,
Conversion of a chaperonin GroEL-independent protein into an obligate substrate.
J Biol Chem 289, 32073-32080 (2014)
-Koike-Takeshita A, Mitsuoka K, *Taguchi H,
Asp52 in combination with Asp398 plays a critical role in ATP hydrolysis of chaperonin GroEL.
J Biol Chem 289, 30005-30011 (2014)
-Niwa, T., Kanamori T., Ueda, T.*, Taguchi, H.*
Global Analysis of Chaperone Effects Using a Reconstituted Cell-Free Translation System
Proc. Natl. Acad. Sci. U.S.A. 109, 8937-8942 (2012)
-Fujiwara, K., Ishihama, Y., Nakahigashi, K., Soga, T. and Taguchi, H.*
A systematic survey of in vivo obligate chaperonin-dependent substrates.
EMBO J. 29, 1552-1564 (2010)
-#Ueno, T, #Taguchi, H, Tadakuma, H., *Yoshida, M, *Funatsu, T. [# equally contributed]
GroEL mediates protein folding with a two successive timer mechanism.
Mol. Cell 14, 423-434 (2004)
-Taguchi, H., Ueno, T., Tadakuma, H., *Yoshida, M., *Funatsu, T.
Single-molecule observation of protein-protein interactions in the chaperonin system.
Nat. Biotechnol. 19, 861-865 (2001)
-Taguchi, H., Konishi, J., Ishii, N., and *Yoshida, M.
A chaperonin from a thermophilic bacterium, Thermus thermophilus, that controls refoldings of several thermophilic enzymes.
J Biol Chem 266, 22411-22418 (1991)