Research summary
Life depends on the function of proteins. Since the function of proteins is fundamentally dependent on their three-dimensional structure, understanding how proteins are synthesized and folded into the correct tertiary structure is essential for understanding life. Proteins are chains of amino acids (polypeptides), and the blueprint of how the 20 amino acids line up to form a polypeptide is encoded in the genes, which consist of DNA. Although the whole genomes of many organisms, including human, have been fully decoded, our understanding of how proteins fold is still incomplete.
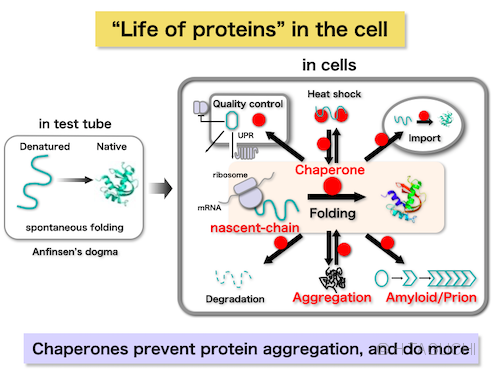
More than 50 years ago, Christian Anfinsen demonstrated that protein folding is a spontaneous process. In other words, amino acid sequence determines the unique tertiary structure. However, protein folding does not always proceed spontaneously in the cell. Protein folding sometimes fails, resulting in aggregate formation. Aggregate formation is notorious not only to impair the protein function, but also to be associated with many human diseases such as Alzheimer’s disease.
In the cell protein folding is assisted by caretaker proteins, called molecular chaperones. Chaperones assist the folding by preventing the formation of aggregates. First definition of chaperones is “a protein family that assists the protein folding but is not included in the final structure”. After the decades of study, we now know that chaperones are widely important to maintain the protein homeostasis (proteostasis) in the cell. Chaperones are essential from the birth to the death of protein in the cell (“life of proteins”).
In our laboratory, we are interested in elucidating the molecular mechanism of chaperones, prions, and translation dynamics in the cell.