Prion
Prion: self-propagating protein conformations
【Concept of prions】
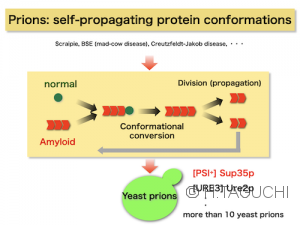
Prion is a proteinaceous infectious factor. As shown in the figure, the abnormal conformation of the protein (this is the prion) autocatalytically transforms the normal conformation into the abnormal state. The “abnormal” form of the protein is called amyloid, in which proteins orderly assemble each other via intermolecular beta-sheets, eventually forming fibrillar structures, amyloids. The concept of prions was proposed by Prusiner to explain the infection mechanism of mammalian neurodegenerative diseases (e.g., scrapie in sheep, Creutzfeldt-Jakob disease, mad cow disease, etc.). The prion is an interesting biological phenomenon since the abnormal conformation propagates and transmits via a “proteins-only” mechanism.
【Prion and protein science】
It is common knowledge in the central dogma of biology that proteins are final products and do not reproduce on their own. However, in prions, a particular tertiary structure of the protein is autocatalytically propagated. Prions are usually considered as fatal human diseases such as mad cow disease and Creutzfeldt-Jakob disease, but are also intriguing unconventional phenomenon from the standpoint of protein science.
【Yeast prions: expansion of the prion concept】
In addition, the prion-like behavior of protein structures is not limited to mammalian prions, but has spread to other phenomena as well. A typical example is the yeast prions. Thanks to the tractable model organism of yeast, research on budding yeast prions, which is difficult to do with mammalian prions, has progressed, and our understanding of the prion phenomenon has advanced. Furthermore, the generality of the prion phenomenon and its biological significance have been discussed.
【Our interest】
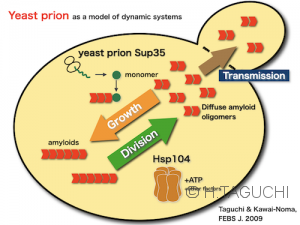
Using yeast prions as a model, our laboratory has conducted various studies to explain the mechanisms of prion propagation at molecular levels. In particular, we have been studying how yeast prions form ordered aggregates (fibrils) in vitro, and how they grow and propagate in living yeast cells. In particular, we have been trying to directly visualize how prions are propagated in living yeast cells.
Recently, yeast prion project is not popular in our lab since the basic mechanism of yeast prion has been clarified after the extensive studies for decades. However, recently it has been reported that Sup35 protein, as a model yeast prion, undergoes liquid-liquid phase separation and forms droplets under nutrient starvation conditions. Therefore, it is expected that there will be interesting findings not only in the monomer-amyloid transition but also in the phase separation depending on the cellular conditions.
【Selected publications】
-†Konno H, †Watanabe-Nakayama T, Uchihashi T, Okuda M, Zhu L, Kodera N, Kikuchi Y, *Ando T, *Taguchi, H. † equally contributed authors
Dynamics of oligomer and amyloid fibril formation by yeast prion Sup35 observed by high-speed atomic force microscopy.
Proc Natl Acad Sci USA 117, 7831-7836 (2020)
-Kawai-Noma, S., Pack, C-G., Kojidani, T., Asakawa, H., Hiraoka, Y., Kinjo, M., Haraguchi, T., *Taguchi H, and Hirata, A.
In vivo evidence for the fibrillar structures of Sup35 prions in yeast cells.
J Cell Biol 190, 223-231 (2010)
-*Taguchi, H. and Kawai-Noma, S.
Diffuse oligomer-based transmission of yeast prions. (Review)
FEBS J 277, 1359-1368 (2010)
-Inoue, Y., Taguchi, H., Kishimoto, A., *Yoshida, M.
Hsp104 binds to yeast sup35 prion fiber but needs other factor(s) to sever it.
J Biol Chem 279, 52319-52323 (2004)
-Inoue, Y., Kishimoto, A., Hirao, J., *Yoshida, M, Taguchi, H.
Strong growth polarity of yeast prion-fiber revealed by single fiber imaging.
J Biol Chem 276, 35227-35230 (2001)