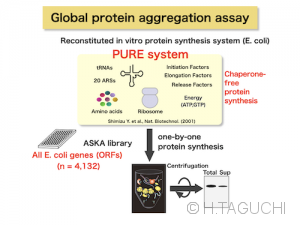
Nascent-chain biology
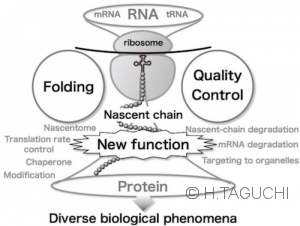
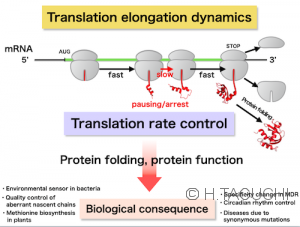
【Concept of Nascent-chain biology】Life depends on correct gene expressions. Proteins do not instantaneously finish the synthesis and folding into functioning products, but experience the nascent peptidyl-tRNAs, defined as “nascent chains”, during the translation. So far, nascent chains are regarded as transient intermediates during the protein synthesis. However, recent advances have revealed that nascent chains are directly involved in a variety of cellular processes including self-maturation and the quality control system of protein and mRNA. In addition, the dysfunction of the maturation and the quality control system could perturb cellular homeostasis, often leading to human diseases.
The concept of nascent chains joins the protein and RNA researchers to generate a new field called “Nascent-chain biology”. Our project aims to understand the roles of the “nascent chains” in the gene expression and cellular homeostasis.
 |
 |
【Selected publications】
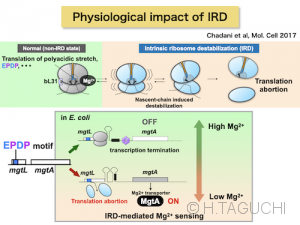
-Chadani Y, Niwa T, Izumi T, Sugata N, Nagao A, Suzuki T, Chiba S, *Ito K, *Taguchi H.
Intrinsic ribosome destabilization underlies translation and provides an organism with a strategy of environmental sensing.
Mol Cell 68, 528-539 (2017)
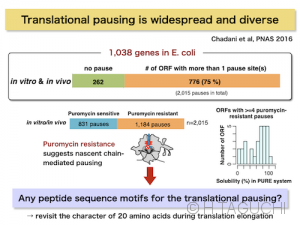
-Chadani Y, Niwa T, Chiba S, *Taguchi H, *Ito K.
Integrated in vivo and in vitro nascent chain profiling reveals widespread translational pausing.
Proc Natl Acad Sci USA 113, E829-38 (2016)